(Peer-Reviewed) Essential role of MALAT1 in reducing traumatic brain injury
Na Wu ¹, Chong-Jie Cheng 程崇杰 ¹, Jian-Jun Zhong 钟建军 ¹, Jun-Chi He 何骏驰 ¹, Zhao-Si Zhang 张兆斯 ¹, Zhi-Gang Wang ¹, Xiao-Chuan Sun 孙晓川 ¹, Han Liu 刘汉 ¹ ²
¹ Department of Neurosurgery, The First Affiliated Hospital of Chongqing Medical University, Chongqing, China
中国 重庆 重庆医科大学附属第一医院神经外科
² Department of Neurosurgery, Qilu Hospital of Shandong University (Qingdao Campus), Qingdao, Shandong Province, China
中国 山东 青岛 山东大学齐鲁医院(青岛)神经外科
Abstract
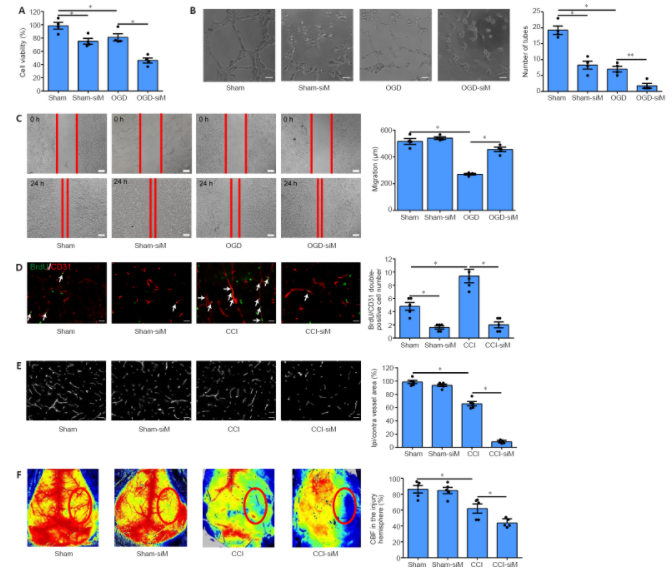
As a highly evolutionary conserved long non-coding RNA, metastasis associated lung adenocarcinoma transcript 1 (MALAT1) was first demonstrated to be related to lung tumor metastasis by promoting angiogenesis. To investigate the role of MALAT1 in traumatic brain injury, we established mouse models of controlled cortical impact and cell models of oxygen-glucose deprivation to mimic traumatic brain injury in vitro and in vivo.
The results revealed that MALAT1 silencing in vitro inhibited endothelial cell viability and tube formation but increased migration. In MALAT1-deficient mice, endothelial cell proliferation in the injured cortex, functional vessel density and cerebral blood flow were reduced. Bioinformatic analyses and RNA pull-down assays validated enhancer of zeste homolog 2 (EZH2) as a downstream factor of MALAT1 in endothelial cells.
Jagged-1, the Notch homolog 1 (NOTCH1) agonist, reversed the MALAT1 deficiency-mediated impairment of angiogenesis. Taken together, our results suggest that MALAT1 controls the key processes of angiogenesis following traumatic brain injury in an EZH2/NOTCH1-dependent manner.
A 4096-element 3D-integrated Si-SiN optical phased array for high-power coherent LiDAR
Han Wang, Weimin Xie, Xin Yan, Jiaqi Li, Youxi Lu, Ping Jiang, Feng Li, Kai Jin, Xu Yang, Jiali Jiang, Keran Deng, Weishuai Chen, Jing Luo, Li Jin, Junbo Feng, Kai Wei
Opto-Electronic Technology
2026-03-20
High-speed and large-capacity visible light communication for 6G: advances and perspectives
Nan Chi, Zhilan Lu, Fujie Li, Haoyu Zhang, Yunkai Wang, Xinyi Liu, Zhiwu Chen, Zhe Feng, Zhuoran Hu, Zhixue He, Ziwei Li, Chao Shen, Junwen Zhang
Opto-Electronic Technology
2026-03-20
Holotomography-driven learning unlocks in-silico staining of single cells in flow cytometry by avoiding fluorescence co-registration
Daniele Pirone, Giusy Giugliano, Michela Schiavo, Annalaura Montella, Martina Mugnano, Vincenza Cerbone, Maddalena Raia, Giulia Scalia Ivana Kurelac, Diego Luis Medina, Lisa Miccio Mario Capasso, Achille Iolascon, Pasquale Memmolo, Pietro Ferraro
Opto-Electronic Science
2026-02-25
A hybrid integrated high-precision tunable semiconductor laser
Yiran Zhu, Botao Fu, Zhiwei Fang, Qiyue Hu, Jianping Yu, Yunpeng Song, Yu Ma, Min Wang, Kunpeng Jia, Zhenda Xie, Ya Cheng
Opto-Electronic Advances
2026-02-12