(Peer-Reviewed) From aniline to phenol: carbon-nitrogen bond activation via uranyl photoredox catalysis
Deqing Hu ¹, Yilin Zhou ¹, Xuefeng Jiang 姜雪峰 ¹ ² ³
¹ Shanghai Key Laboratory of Green Chemistry and Chemical Process, School of Chemistry and Molecular Engineering, East China Normal University, Shanghai 200062, China
中国 上海 华东师范大学化学与分子工程学院,上海市绿色化学与化工过程绿色化重点实验室
² State Key Laboratory of Organometallic Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai 200032, China.
中国 上海 中国科学院上海有机化学研究所金属有机化学国家重点实验室
³ State Key Laboratory of Elemento-Organic Chemistry, Nankai University, Tianjin 300071, China
中国 天津 南开大学元素有机化学国家重点实验室
National Science Review, 2021-08-20
Abstract
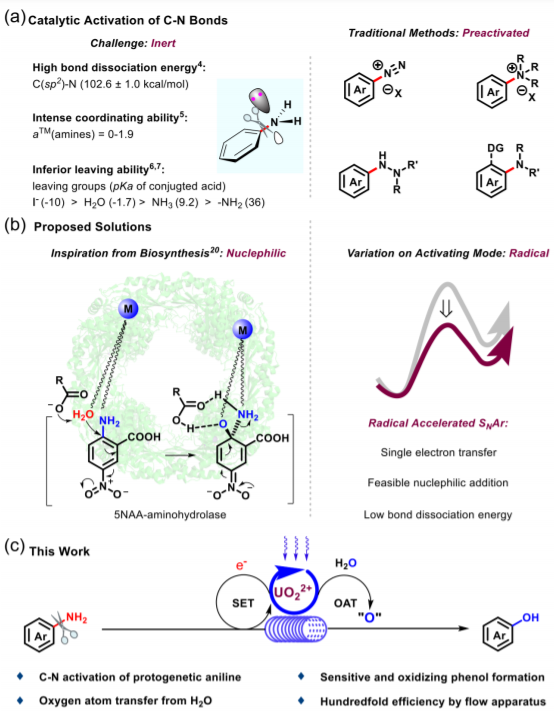
Carbon-nitrogen bond activation, via uranyl photoredox catalysis with water, enabled the conversion from 40 protogenetic anilines, 8 N-substituted anilines, and 9 aniline-containing natural products/pharmaceuticals to the corresponding phenols at ambient environment. Single electron transfer process between protonated aniline and uranyl catalyst, which was disclosed by radical quenching experiments and Stern-Volmer analysis, facilitated the following oxygen atom transfer process between radical cation of protonated anilines and uranyl peroxide originating from water-splitting.
¹⁸O labelling and ¹⁵N tracking unambiguously depicted that the oxygen came from water and amino group leaved as ammonium salt. Hundredfold efficiency of flow operation demonstrated the great potential of the conversion process in industrial synthetic application.
A hybrid integrated high-precision tunable semiconductor laser
Yiran Zhu, Botao Fu, Zhiwei Fang, Qiyue Hu, Jianping Yu, Yunpeng Song, Yu Ma, Min Wang, Kunpeng Jia, Zhenda Xie, Ya Cheng
Opto-Electronic Advances
2026-02-12
Millisecond-level electrically switchable metalens for adaptive rotational depth mapping and diffraction-limited imaging
Yeseul Kim, Jihae Lee, Won-Sik Kim, Hyeonsu Heo, Dongmin Jeon, Beomha Yang, Xiaotong Li, Harit Keawmuang, Shiqi Hu, Young-Ki Kim, Trevon Badloe, Junsuk Rho
Opto-Electronic Advances
2026-02-12
Integrated metasurface-freeform system enabled multi-focal planes augmented reality display
Shifei Zhang, Lina Gao, Yidan Zhao, Yongdong Wang, Bo Wang, Junjie Li, Jiaxi Duan, Dewen Cheng, Cheng-Wei Qiu, Yongtian Wang, Tong Yang, Lingling Huang
Opto-Electronic Science
2026-01-23