(Peer-Reviewed) All-climate aqueous Na-ion batteries using “Water-in-Salt” electrolyte
Yu Zhang ¹, Jie Xua ¹, Zhi Li ¹, Yanrong Wang ¹, Sijia Wang ², Xiaoli Dong 董晓丽 ¹, Yonggang Wang 王永刚 ¹
¹ Department of Chemistry and Shanghai Key Laboratory of Molecular Catalysis and Innovative Materials, Institute of New Energy, iChEM (Collaborative Innovation Center of Chemistry for Energy Materials), Fudan University, Shanghai 200433, China
中国 上海复旦大学化学系、上海市分子催化和功能材料重点实验室、复旦大学新能源研究院、能源材料化学协同创新中心
² College of Pharmacy, Henan University of Chinese Medicine, Zhengzhou 450046, China
中国 郑州 河南中医药大学药学院
Science Bulletin, 2021-08-17
Abstract
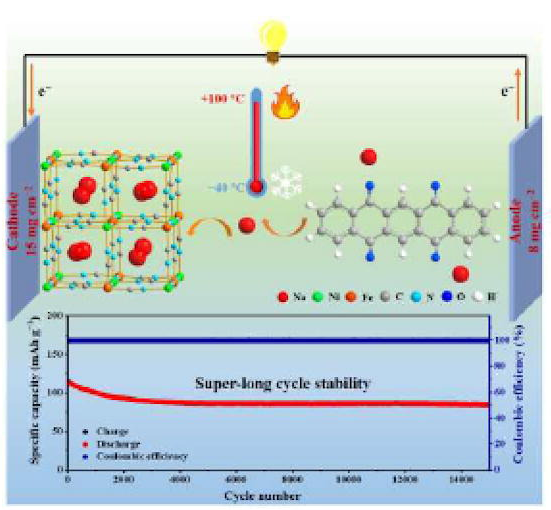
Aqueous Na-ion batteries have been extensively studied for large-scale energy storage systems. However, their wide application is still limited by their inferior cycle stability (< 3000 cycles) and poor temperature tolerance. Furthermore, many of the reported high rate behaviors are achieved at a low mass loading (<3 mg cm−2) of the electrodes. Herein, we propose an aqueous Na-ion battery which includes a Ni-based Prussian blue (NiHCF) cathode, a carbonyl-based organic compound, 5,7,12,14-pentacenetetrone (PT) anode and a “water-in-salt” electrolyte (17 mol kg−1 NaClO4 in water).
Its operation involves the reversible coordination reaction of the PT anode and the extraction/insertion of Na+ in the NiHCF cathode. It is demonstrated that the wide internal spaces of the PT anode and NiHCF cathode can not only buffer the volumetric change induced by Na+ storage, but also enable fast kinetics.
The full cell exhibits a supercapacitor-like rate performance of 50 A g−1 (corresponding to a discharge or charge within 6.3 s) and a super-long lifespan of 15,000 cycles. Moreover, the excellent rate performance can still be preserved even with a high mass loading of the electrodes (15 mgNiHCF cm−2 and 8 mgPT cm−2). Especially, the cell can work well in a wide temperature range, from −40 to 100 °C, showing a typical all-climate operation.
A 4096-element 3D-integrated Si-SiN optical phased array for high-power coherent LiDAR
Han Wang, Weimin Xie, Xin Yan, Jiaqi Li, Youxi Lu, Ping Jiang, Feng Li, Kai Jin, Xu Yang, Jiali Jiang, Keran Deng, Weishuai Chen, Jing Luo, Li Jin, Junbo Feng, Kai Wei
Opto-Electronic Technology
2026-03-20
High-speed and large-capacity visible light communication for 6G: advances and perspectives
Nan Chi, Zhilan Lu, Fujie Li, Haoyu Zhang, Yunkai Wang, Xinyi Liu, Zhiwu Chen, Zhe Feng, Zhuoran Hu, Zhixue He, Ziwei Li, Chao Shen, Junwen Zhang
Opto-Electronic Technology
2026-03-20
Holotomography-driven learning unlocks in-silico staining of single cells in flow cytometry by avoiding fluorescence co-registration
Daniele Pirone, Giusy Giugliano, Michela Schiavo, Annalaura Montella, Martina Mugnano, Vincenza Cerbone, Maddalena Raia, Giulia Scalia Ivana Kurelac, Diego Luis Medina, Lisa Miccio Mario Capasso, Achille Iolascon, Pasquale Memmolo, Pietro Ferraro
Opto-Electronic Science
2026-02-25