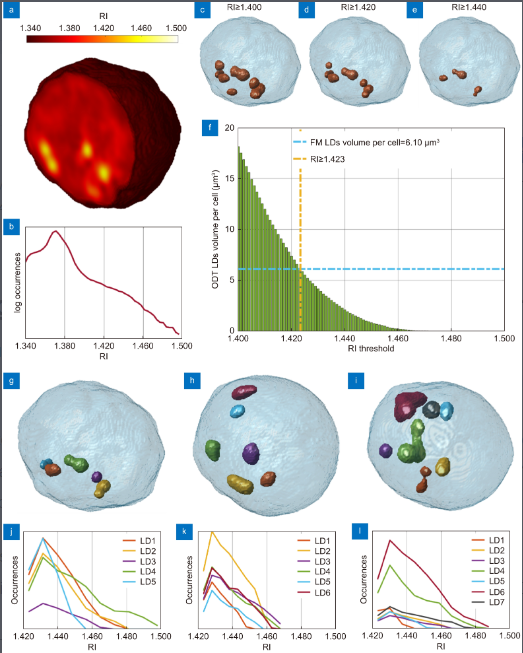
(Peer-Reviewed) 3D imaging lipidometry in single cell by in-flow holographic tomography
Daniele Pirone ¹ ², Daniele Sirico ¹ ³, Lisa Miccio ¹, Vittorio Bianco ¹, Martina Mugnano ¹, Danila del Giudice ¹ ⁴, Gianandrea Pasquinelli ⁵ ⁶, Sabrina Valente ⁵, Silvia Lemma ⁷ ⁸ ⁹, Luisa Iommarini ⁹ ¹⁰, Ivana Kurelac ⁷ ⁸ ⁹, Pasquale Memmolo ¹, Pietro Ferraro ¹
¹ CNR-ISASI, Institute of Applied Sciences and Intelligent Systems “E. Caianiello”, Via Campi Flegrei 34, Pozzuoli, Napoli 80078, Italy
² Department of Electrical Engineering and Information Technologies, University of Naples “Federico II”, via Claudio 21, Napoli 80125, Italy
³ Department of Chemical, Materials and Production Engineering of the University of Naples Federico II, Piazzale Tecchio 80, Napoli 80125, Italy
⁴ Department of Mathematics and Physics, University of Campania “Luigi Vanvitelli”, Caserta 81100, Italy
⁵ Biotechnology and Methods in Laboratory Medicine, Department of Experimental, Diagnostic and Specialty Medicine (DIMES), University of Bologna, Bologna 40126, Italy
⁶ Subcellular Nephro-Vascular Diagnostic Program, Pathology Unit S.Orsola IRCCS, University of Bologna, Bologna 40126, Italy
⁷ Unit of Medical Genetics, Department of Medical and Surgical Sciences (DIMEC), University of Bologna, Via Massarenti 9, Bologna 40138, Italy
⁸ Study and Research Center on Gynecological Neoplasias, Department of Medical and Surgical Sciences (DIMEC), University of Bologna, Via Massarenti 9, Bologna 40138, Italy
⁹ Center for Applied Biomedical Research (CRBA), University of Bologna, Bologna 40138, Italy
¹⁰ Department of Pharmacy and Biotechnology (FABIT), University of Bologna, Bologna 40126, Italy
Opto-Electronic Advances, 2022-08-25